Biophysics, Dendrites, Neural Computation
How do the biophysical features of neurons give rise to the computational power of the brain?
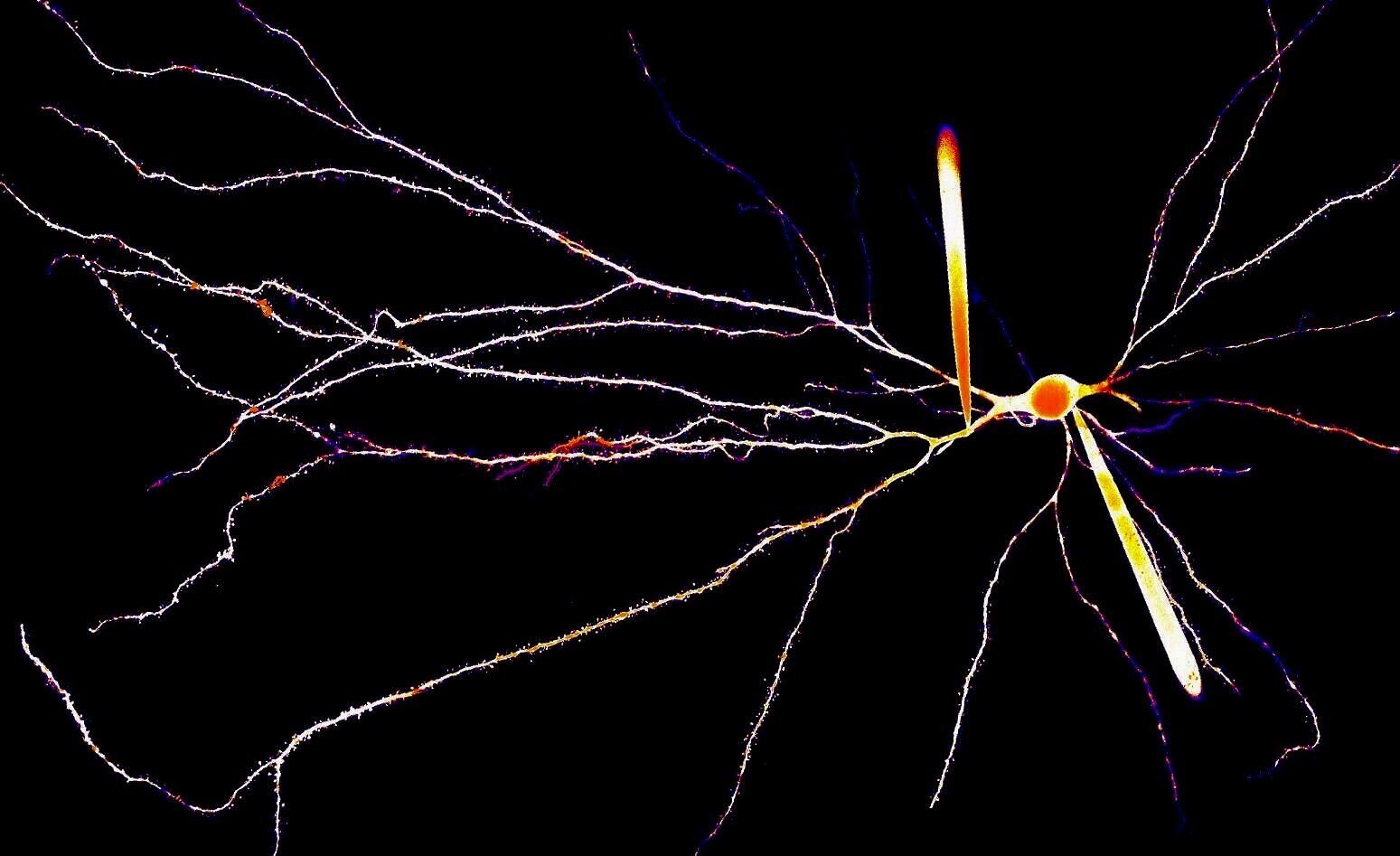
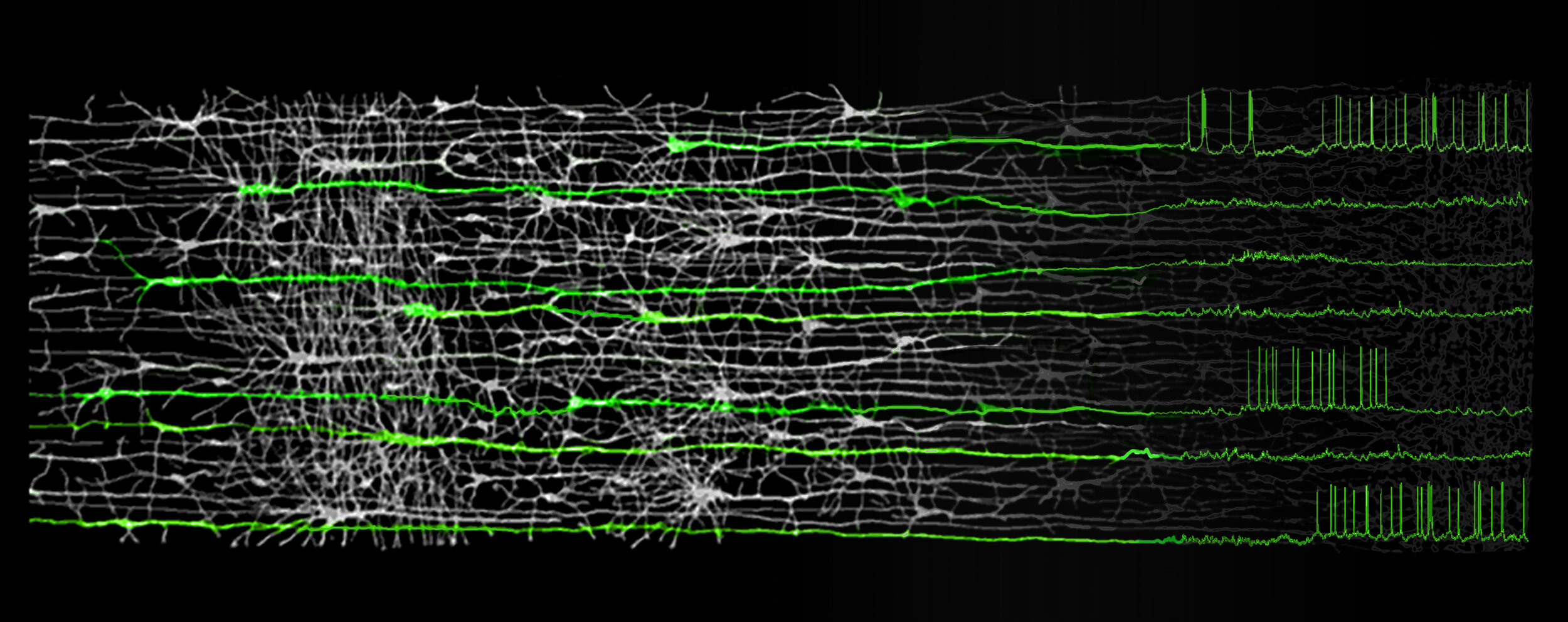
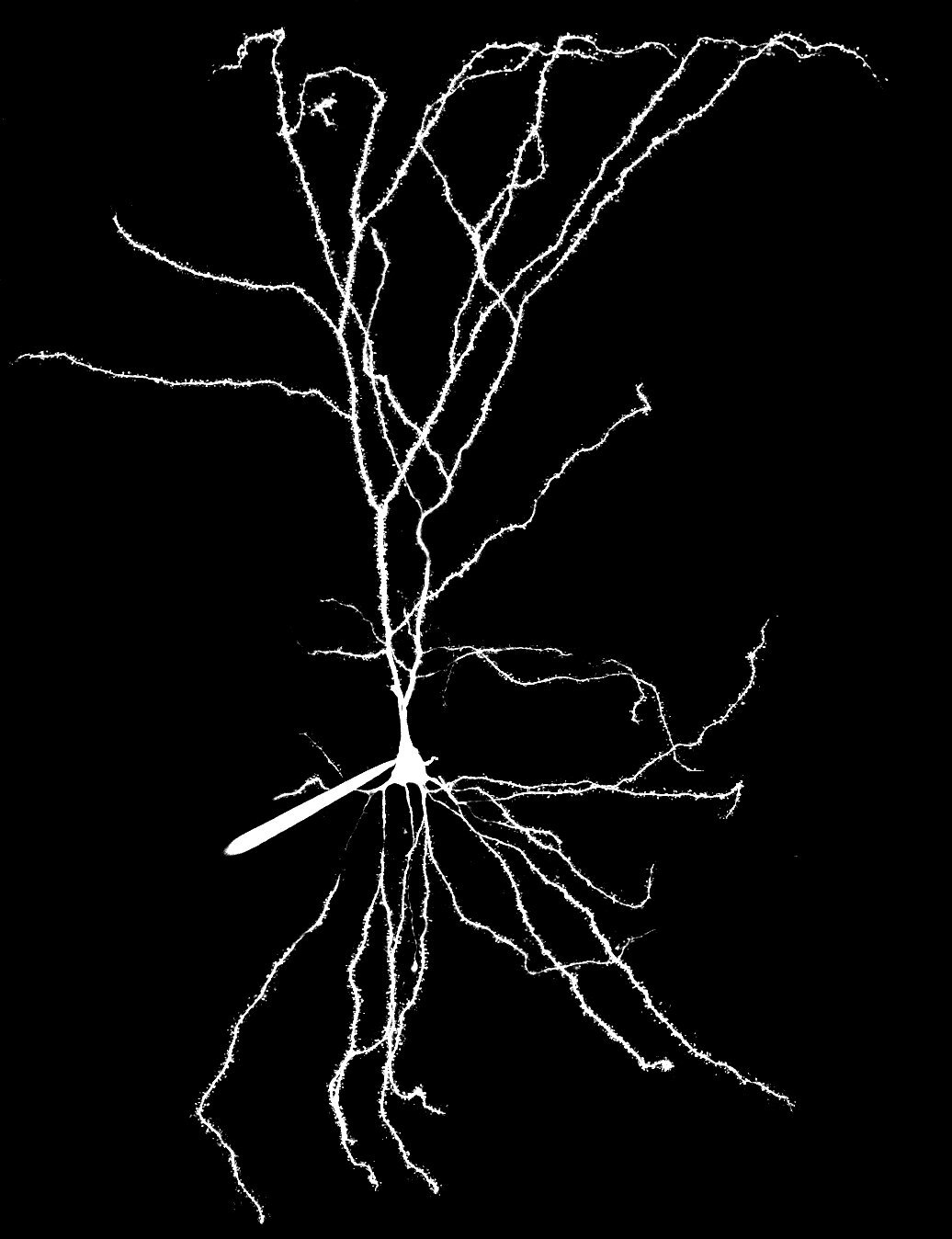
Our laboratory studies how the biophysical features of individual neurons endow neural circuits with powerful processing capabilities, ultimately facilitating the complex computations required to drive adaptive behavior. A principal focus of our work is the role of dendrites, the elaborate tree-like structures where neurons receive the vast majority of afferent input. The spatial arrangement of synaptic contacts on dendrites and the interaction of various biophysical mechanisms enable complex integration of synaptic inputs – our hypothesis is that circuit-level computations are built out of these fundamental operations.
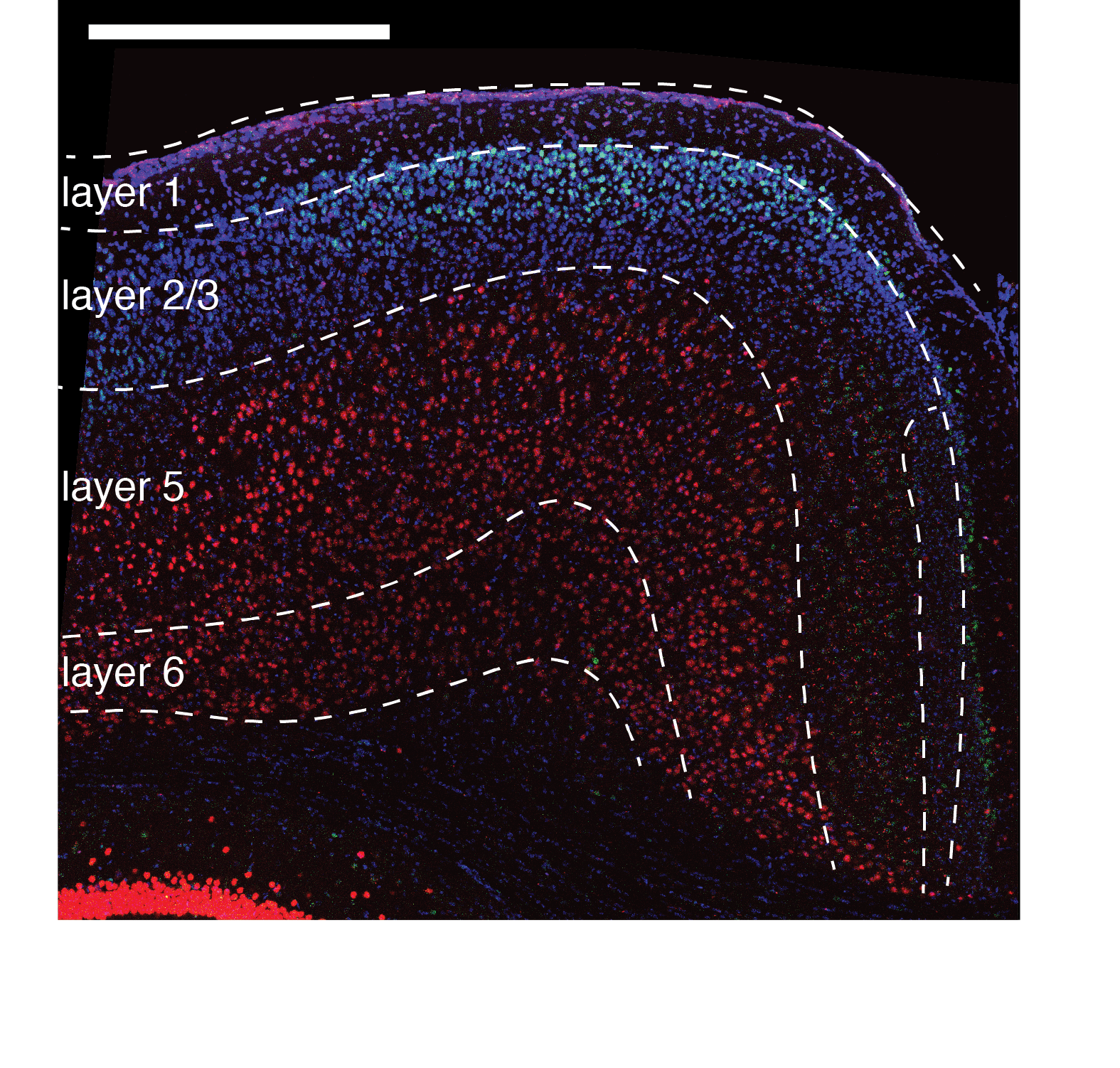
Statement on Diversity and Inclusion
The Harnett lab is an inclusive and collaborative work environment where members are empowered to freely and openly share their different views and ideas. Our lab's strength arises directly from these diverse perspectives. As such, we strive to ensure that all who come into contact with our lab are treated with respect and dignity. Working in the international world of academia means continuously learning and adapting to meet this goal.
We are aligned with the mission statements of MIT and BCS in terms of standing against discrimination, racism, and bias. We further believe it is important to establish our own clear expectations and standards. To this end, and to foment a respectful and friendly work environment, we expect lab members to educate themselves on common workplace issues that arise from bias and privilege. This is a continuing subject of discussion in our lab as we all strive towards our shared goals of scientific excellence, inclusivity, equity, and personal fulfillment.